 
Testing for anti-double-stranded DNA and antineutrophil cytoplasmic autoantibodies (ANCA) was negative. In the absence of other ANA and negative PM-Scl in a control measurement these findings were interpreted as an unspecific reaction. ANA differentiation detected antibodies against U1-RNP and PM-Scl. In immunoblot, antinuclear antibodies (ANA) were positive. Coombs test, cold agglutinins, hemoglobin electrophoresis and glucose-6-phosphate dehydrogenase activity showed unremarkable results. 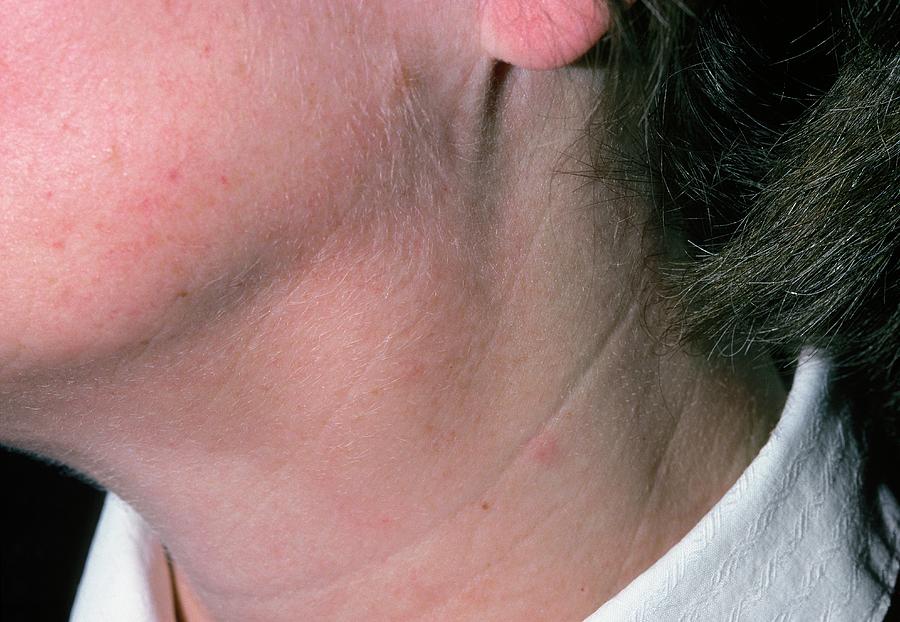
The only remarkable value in the serological screening was an increase in Mycoplasma pneumonia IgG (22.3, reference < 9) and IgM (15.9, reference < 9) by enzyme immunoassay, though ten days later, no significant changes in antibody titers were observed. Serological and PCR virus tests (EBV, CMV, hepatitis B, C, E, HIV, HSV, Parvo-B19) were negative. No additional lymphadenopathy or pulmonary infiltrates were detected. An abdominal ultrasound revealed a splenomegaly (158 × 57 mm), while a computed tomography chest scan confirmed enlarged cervical and supraclavicular lymph nodes with a maximum diameter of 19 × 10 mm. Serum creatinine levels were normal at all times. Subsequent laboratory testing on day 19 showed a further decrease of the WBC count (1.95 × 10^9/l), an increase of LDH (1184 U/l) and liver function parameters (ASAT 162 U/l, ALAT 121 U/l, GGT 40 U/l), as well as decreased haptoglobin (< 0,1 g/l) (Fig. The patient was in a reduced general condition with a painful cervical and supraclavicular bilateral lymphadenopathy. At the time of presentation at the emergency department on day 16, laboratory testing revealed a reduced total white blood cell count (WBC, 2.4 × 10^9/l), elevated lactate dehydrogenase (LDH, 904 U/l), and slightly elevated aspartate aminotransferase (AST, 72 U/l). After a slight improvement of symptoms, she again developed fever, chills, increasing weakness, and nausea from day 13 after vaccination. 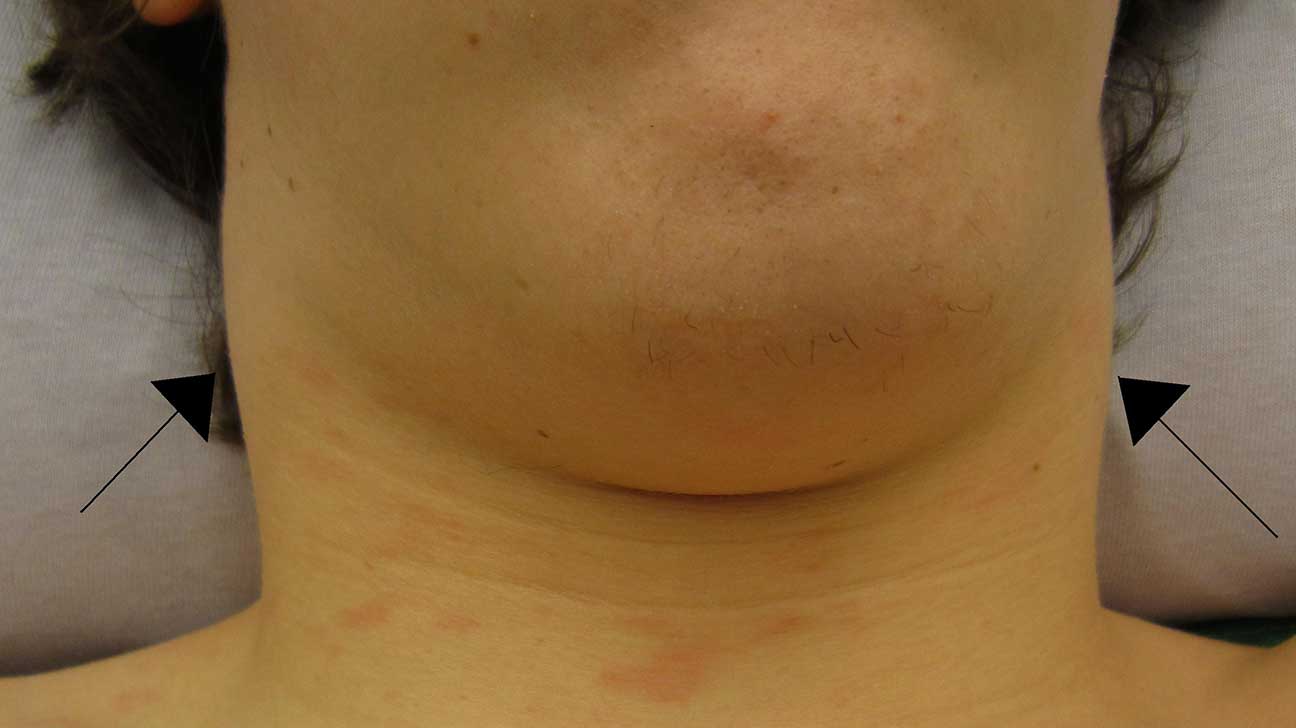
A 24-year-old, white female with no remarkable medical or travel history developed fever and unspecific fatigue for ten days after the first COVID-19 vaccination with Comirnaty. Even with current treatment options, it has a 50% lethality. HLH can rapidly progress to multiple organ failure and, if untreated, is often fatal. HLH is a life-threatening hyperinflammatory syndrome caused by aberrantly activated macrophages and cytotoxic T cells. ConclusionĬOVID-19 vaccines may occasionally trigger HLH, and Anakinra may be an efficacious treatment option for this condition. Eight patients underwent combination treatment. Treatment modalities included corticosteroids ( n = 13), Anakinra ( n = 5), IVIGs ( n = 5), and etoposide ( n = 2). The addition of the human interleukin 1 receptor antagonist Anakinra resulted in full recovery within 6 weeks after vaccination.Ī literature search revealed 15 additional cases of HLH after SARS-CoV-2 vaccination, the majority after immunization with Comirnaty ( n = 7) or the viral vector vaccine Vaxzevria ( n = 6). The patient was initially treated with intravenous immunoglobulins (IVIGs) and dexamethasone without response. Diagnosis was made according to HLH-2004 criteria the HScore was 259 (> 99% HLH probability) with maximum ferritin of 138.244 µg/L. ResultsĪ 24-year-old female developed HLH after immunization with the mRNA COVID-19 vaccine Comirnaty. We report a case of severe hemophagocytic lymphohistiocytosis (HLH) after SARS-CoV-2 immunization and performed a literature search for all reported cases of COVID-19 vaccine-associated HLH. Until now, more than 4.7 billion persons have been vaccinated around the world, and adverse effects not observed in pre-authorization trials are being reported at low frequency. The coronavirus disease 2019 (COVID-19) pandemic has led to the approval of novel vaccines with different mechanisms of action. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
